
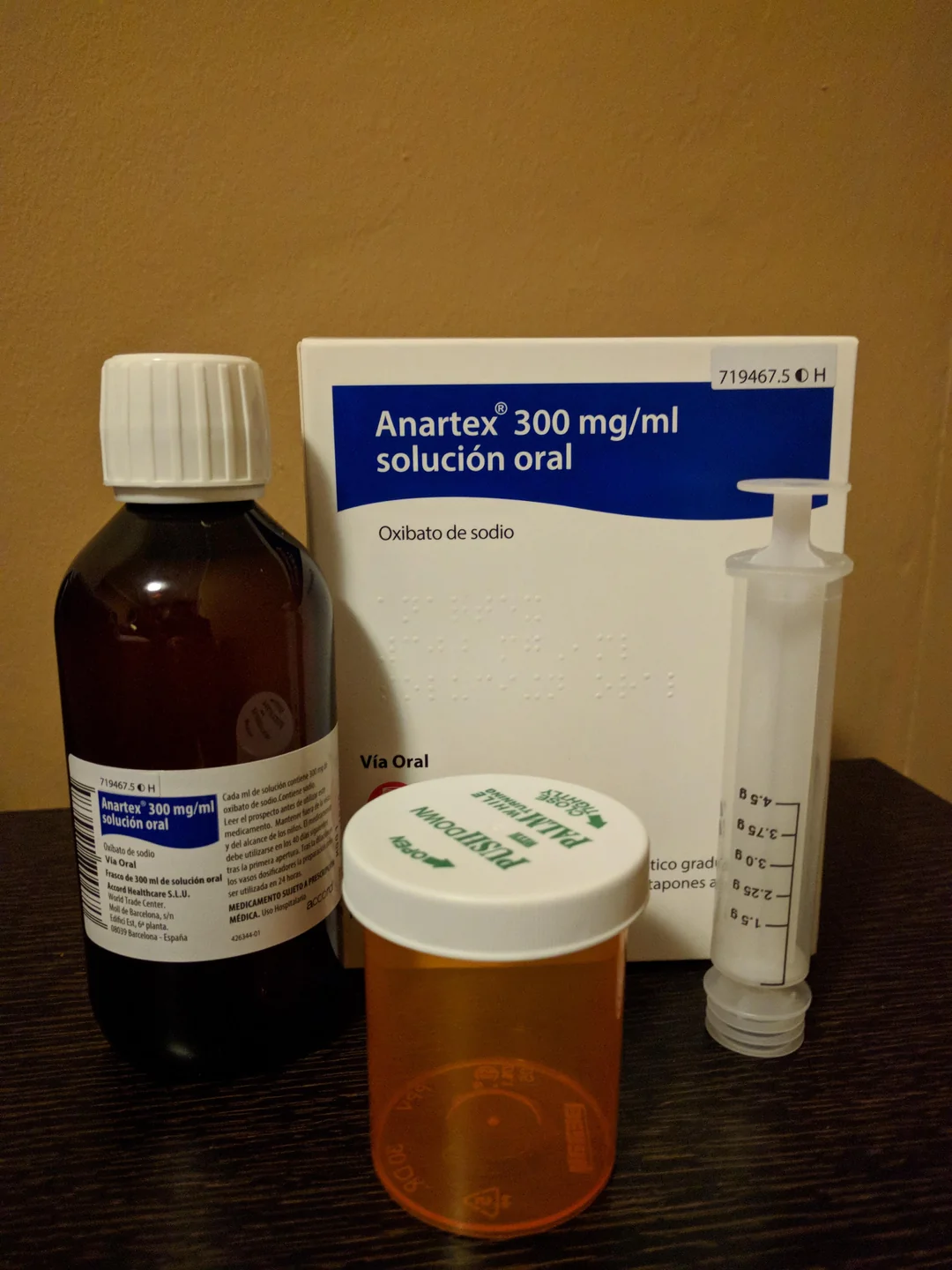
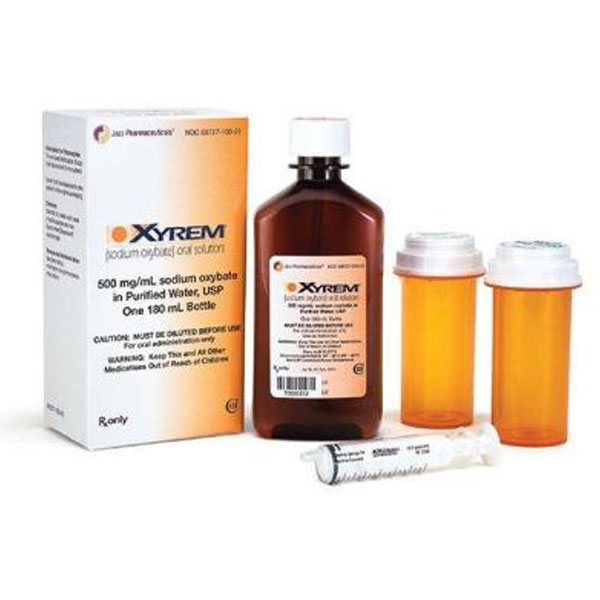
Xyrem (soduim oxybate)180 ml, 500 mg γ-hydroxybutyric acid (GHB) per ml
255 $ – 2 ,000 $Price range: 255 $ through 2 ,000 $
Xyrem 180 ml – 500 mg/ml Sodium Oxybate – Complete Educational Guide
Xyrem (sodium oxybate) is a prescription-only central nervous system (CNS) depressant approved for treating narcolepsy symptoms in patients aged 7 years and older. The standard 180 ml bottle contains an oral solution with 500 mg sodium oxybate per ml (0.5 g/ml), totaling 90 grams of active ingredient (90,000 mg). This page provides factual, evidence-based information drawn from official FDA prescribing information, EMA product characteristics, and current medical guidelines (as of 2026).
Critical Reminder: Xyrem is not available for purchase without a valid prescription. It is a Schedule III controlled substance in the United States and a narcotics-regulated medication (BtMG Anlage III) in Germany and the EU. Any non-prescription offers are illegal and carry extreme health risks due to counterfeit products, incorrect dosing, or contamination.
What Is Xyrem? (Sodium Oxybate Oral Solution)
Xyrem is an FDA- and EMA-approved medication containing sodium oxybate, the sodium salt of gamma-hydroxybutyric acid (GHB). It works by consolidating nighttime sleep, thereby reducing cataplexy and excessive daytime sleepiness in narcolepsy patients. The exact mechanism is not fully understood but involves modulation of GABA-B receptors and other neurotransmitter systems.
- Concentration: 500 mg sodium oxybate per ml (equivalent to ~413 mg/ml oxybate base)
- Bottle details: 180 ml fill volume in a 240 ml amber PET bottle with child-resistant cap
- Appearance: Clear to slightly opalescent liquid
- Sodium content: High — up to ~1,640 mg sodium at the maximum 9 g nightly dose (important for patients on sodium-restricted diets)
- Inactive ingredients: Purified water, malic acid, sodium hydroxide (pH adjusted to ~7.5)
Xyrem includes dosing accessories: an oral syringe, press-in bottle adapter, and child-resistant dosing cups.
Approved Indications
Xyrem is indicated exclusively for:
- Treatment of cataplexy in patients with narcolepsy
- Treatment of excessive daytime sleepiness (EDS) in patients with narcolepsy
It is approved for adults, adolescents, and children aged 7 years and older in both the US and EU. It is not approved for idiopathic hypersomnia, fibromyalgia, alcohol withdrawal, or any recreational/off-label purposes.
Key Safety Warnings & Boxed Warnings
Xyrem carries the most serious FDA boxed warnings due to life-threatening risks:
- Profound CNS depression — Can cause respiratory depression, coma, and death, even at therapeutic doses
- High abuse and misuse potential — Sodium oxybate is chemically identical to illicit GHB, a known drug of abuse
- Absolute contraindication with alcohol, sedative-hypnotics (benzodiazepines, z-drugs), opioids, or other CNS depressants
- Restricted distribution — Available only through the XYWAV and XYREM REMS Program (US) or equivalent EU risk management systems
Prescribers and patients must enroll in REMS: prescriber certification, patient enrollment, and dispensing only through certified pharmacies.
Common side effects include nausea, dizziness, enuresis (bedwetting), sleep paralysis, confusion, hallucinations, and sleepwalking.
Proper Dosing & Administration (Medical Supervision Only)
Dosing is always individualized by a qualified sleep specialist or neurologist:
- Starting dose: 4.5 g per night (2.25 g at bedtime + 2.25 g 2.5–4 hours later)
- Titration: Increase by 1.5 g per night weekly
- Effective range: 6–9 g per night (maximum studied dose; >9 g not recommended)
- Administration rules:
- Dilute each dose in approximately 60 ml of water
- Take on an empty stomach (at least 2–3 hours after eating)
- Remain in bed immediately after dosing due to rapid and deep sedation
- Set an alarm for the second dose if necessary
Always adhere strictly to the prescribing physician’s instructions and the official package insert.
Storage, Handling & Safe Disposal Guidelines
- Store in the original amber bottle at controlled room temperature (15–30 °C / 59–86 °F)
- Protect from light and keep out of reach of children
- Use the child-resistant cap at all times
- Discard any opened bottle after 90 days (or as indicated on the label)
- Return unused or expired product to a pharmacy for safe disposal — never flush down the toilet or dispose in household trash
- Diluted doses must be consumed within 24 hours
Legality, Availability & REMS Requirements (2026 Update)
Xyrem remains strictly controlled worldwide:
- United States: Schedule III (DEA); requires enrollment in the XYWAV and XYREM REMS Program (prescriber certification, patient enrollment, centralized dispensing)
- European Union / Germany: Prescription-only and BtMG-regulated (Anlage III); no free sale or casual online purchase
- Generic availability: FDA-approved generics (e.g., from Amneal, Ascent, and others) have been available since 2025, but all versions remain subject to REMS-equivalent restrictions
- No legitimate OTC or unrestricted online availability — Only obtainable through enrolled prescribers and certified pharmacies
Products from unverified online sellers or black-market sources are frequently counterfeit, under- or overdosed, or contaminated — creating immediate risk of life-threatening overdose or respiratory arrest.
Why This Educational Page Exists at GHB-GBL
At GHB-GBL, our focus is harm reduction, factual education, and transparency regarding psychoactive and controlled substances. Xyrem (sodium oxybate) is a powerful prescription medication intended solely for the medical treatment of narcolepsy — it is not a recreational compound.
If you or someone you know experiences symptoms of narcolepsy (persistent excessive sleepiness, sudden muscle weakness/cataplexy), seek professional evaluation from a neurologist or sleep medicine specialist. Only qualified medical professionals can diagnose the condition and, if appropriate, prescribe Xyrem through legitimate, REMS-approved channels.
Never attempt to obtain Xyrem outside of a doctor-prescribed, pharmacy-dispensed pathway.
This content is purely informational and based on the latest official FDA and EMA prescribing information (last major updates 2023–2025). It does not constitute medical advice, encourage any illegal activity, or promote non-medical use of any substance. Always consult a licensed healthcare provider and confirm current local regulations.
| Quantity |
20Grams ,25Grams ,50Grams ,100Grams ,250Grams ,500Grams ,1KG |
|---|


Reviews
There are no reviews yet.